
Chapter 1: Measurements and Units
Physical Quantity is a quantity that can be measured. It consists of a numerical magnitude and a unit.
Numbers and Units
Prefixes for SI units
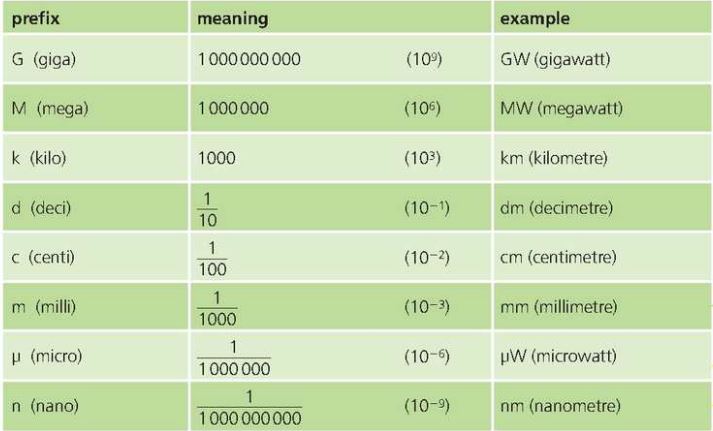
Powers of 10

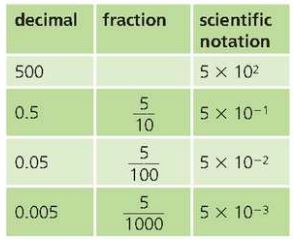
Scientific Notation
Numbers written using powers of ten are in scientific notation or standard form.
For example;
 System of Units
System of Units
SI Units
Are set systematic international units, for measuring mass, time, and other base units and derived quantities.
Mass
Is the measure of the amount of substance in an object.
Has the following effects:
all objects are attracted to the Earth. The greater the mass of an object, the stronger is he Earth’s gravitational pull on it.
all objects resist attempts to make them go faster, slower, or in a different direction. The greater the mass, the greater is the resistance to change in motion.
SI base unit is kilogram (kg).
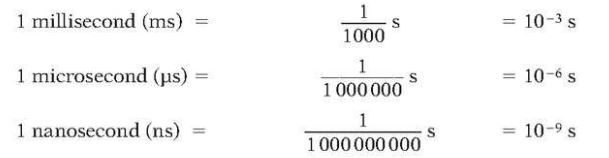
Time
SI base unit is second (s).
Following are some of other units for second:
 Length
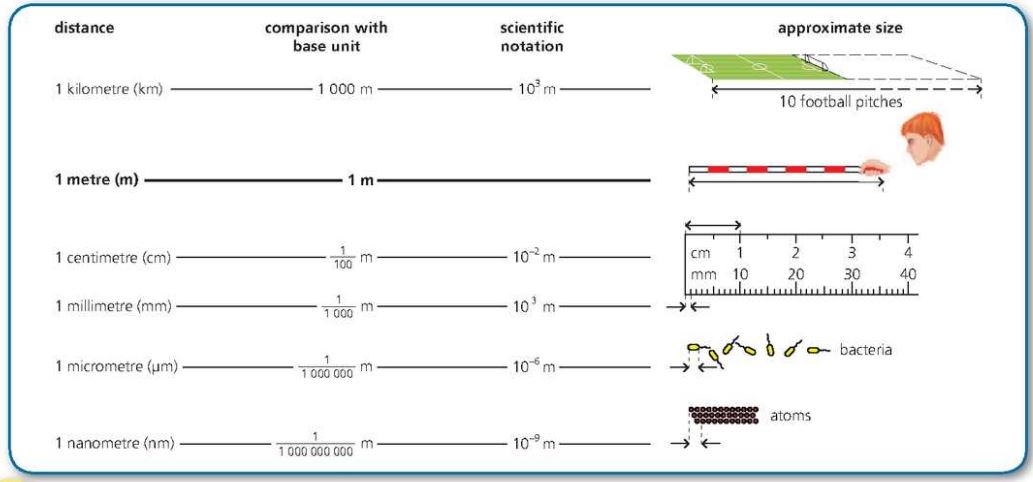
Length
SI base unit is metre (m).
Measuring Length and Time
Measuring Length
Ruler can be used to measure small distances of a few centimetres (cm).
They are able to measure to the nearest millimetre (mm).
Can measure upto one metre.
Example of usage:
height of the table
Micrometer is used to measure objects that are too small to be measured with vernier calipers
Gives a precision of 0.01 mm
Can measure measurements of less than 25 cm.
Example of usage:
diameter of a wire
Vernier Calliper is a useful tool for measuring both internal and external diameters.
They are able to measure to a precision of 0.01 cm.
Measuring Time
Time intervals of many seconds or minutes can be measured using a a stopwatch.
Some instruments have analogue or a digital display.
Volume and Density
Volume
It is the quantity of space an object takes up.
It’s SI unit is cubic metre (m^3).
Density
It is the quantity of mass per unit volume of a substance.
Formula:
Density= Mass/Volume
ρ=m/v
ρ is a greek letter ‘rho’ .
(both equations are same, written in different ways; i.e first one is the word equation and the other one is symbolic equation.
SI unit of density is kilogram per cubic metre (kg m^-3).
Objects that are less dense than water will float on water; and objects that are denser than water will sink.
Measuring Volume and Density
Measuring Volume
Liquid
Its volume can be measured using a measuring cylinder.
Most cylinders have scale marked in millilitres (ml) or cubic centimetres (cm^3).
Regular Solid
If an object has a simple shape, its volume can be calculated through formula. For example:
volume of a regular block = length x width x height
Irregular Solid
If the shape of the object is too awkward to calculate its volume, then immerse the object in a measuring cylinder filled with water and then calculate the increase in water level. This value will be the volume of the object.
Measuring Density
Once volume of the object is obtained through one of the above methods suitable, find the mass of the object using a balance.
Following is one of the example:

Checking the mass and density of a liquid
An easy method of finding the density of a liquid is by using a small float called hydrometer.
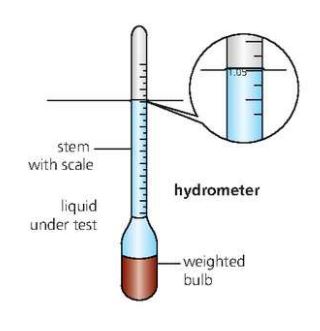
The relative density on a hydrometer indicates the relative density of the liquid.
Checks like these are important in some production processes.
Chapter 1: Measurements and Units
Physical Quantity is a quantity that can be measured. It consists of a numerical magnitude and a unit.
Numbers and Units
Prefixes for SI units

Powers of 10

Scientific Notation
Numbers written using powers of ten are in scientific notation or standard form.
For example;
 System of Units
System of Units
SI Units
Are set systematic international units, for measuring mass, time, and other base units and derived quantities.
Mass
Is the measure of the amount of substance in an object.
Has the following effects:
all objects are attracted to the Earth. The greater the mass of an object, the stronger is he Earth’s gravitational pull on it.
all objects resist attempts to make them go faster, slower, or in a different direction. The greater the mass, the greater is the resistance to change in motion.
SI base unit is kilogram (kg).
Time
SI base unit is second (s).
Following are some of other units for second:
 Length
Length
SI base unit is metre (m).

Measuring Length and Time
Measuring Length
Ruler can be used to measure small distances of a few centimetres (cm).
They are able to measure to the nearest millimetre (mm).
Can measure upto one metre.
Example of usage:
height of the table
Micrometer is used to measure objects that are too small to be measured with vernier calipers
Gives a precision of 0.01 mm
Can measure measurements of less than 25 cm.
Example of usage:
diameter of a wire
Vernier Calliper is a useful tool for measuring both internal and external diameters.
They are able to measure to a precision of 0.01 cm.
Measuring Time
Time intervals of many seconds or minutes can be measured using a a stopwatch.
Some instruments have analogue or a digital display.
Volume and Density
Volume
It is the quantity of space an object takes up.
It’s SI unit is cubic metre (m^3).
Density
It is the quantity of mass per unit volume of a substance.
Formula:
Density= Mass/Volume
ρ=m/v
ρ is a greek letter ‘rho’ .
(both equations are same, written in different ways; i.e first one is the word equation and the other one is symbolic equation.
SI unit of density is kilogram per cubic metre (kg m^-3).
Objects that are less dense than water will float on water; and objects that are denser than water will sink.
Measuring Volume and Density
Measuring Volume
Liquid
Its volume can be measured using a measuring cylinder.
Most cylinders have scale marked in millilitres (ml) or cubic centimetres (cm^3).
Regular Solid
If an object has a simple shape, its volume can be calculated through formula. For example:
volume of a regular block = length x width x height
Irregular Solid
If the shape of the object is too awkward to calculate its volume, then immerse the object in a measuring cylinder filled with water and then calculate the increase in water level. This value will be the volume of the object.
Measuring Density
Once volume of the object is obtained through one of the above methods suitable, find the mass of the object using a balance.
Following is one of the example:

Checking the mass and density of a liquid
An easy method of finding the density of a liquid is by using a small float called hydrometer.

The relative density on a hydrometer indicates the relative density of the liquid.
Checks like these are important in some production processes.
 Knowt
Knowt
