
Chapter 7: Energy for Cells
7.1 Cellular Respiration
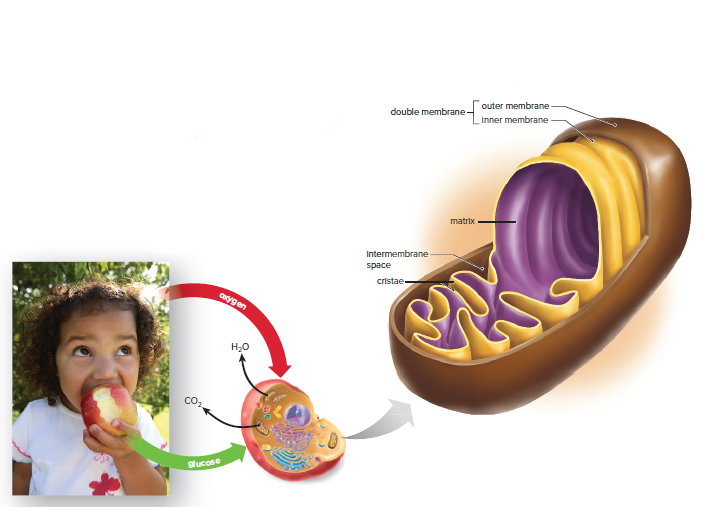
Cellular respiration is the process by which cells produce ATP, the body's main energy source.
ATP is produced by the breakdown of organic molecules, primarily glucose.
Cellular respiration occurs in the mitochondria, the powerhouses of the cell.
Cellular respiration is a redox reaction, which means that electrons are transferred from one molecule to another.
In cellular respiration, glucose is oxidized to form carbon dioxide, and oxygen is reduced to form water.
The energy released from the redox reaction is used to produce ATP.
Phases of Complete Glucose Breakdown
Cellular Respiration
Cellular respiration is a process that cells use to break down glucose and release energy.
The energy released from glucose is used to make ATP, which is the cell's main source of energy.
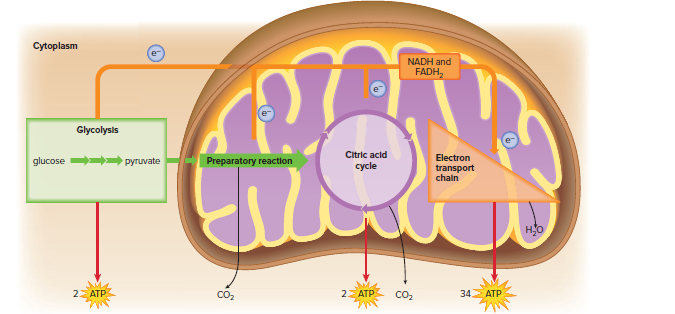
There are four phases of cellular respiration: glycolysis, the preparatory reaction, the citric acid cycle, and the electron transport chain.
Glycolysis
Glycolysis is the first phase of cellular respiration.
It takes place in the cytoplasm of the cell.
In glycolysis, glucose is broken down into two molecules of pyruvate.
Two ATP molecules are produced during glycolysis.
NADH is also produced during glycolysis.
The Preparatory Reaction
The preparatory reaction is the second phase of cellular respiration.
It takes place in the mitochondria.
In the preparatory reaction, pyruvate is broken down into a 2-carbon acetyl group and carbon dioxide.
NADH is also produced during the preparatory reaction.
The Citric Acid Cycle
The citric acid cycle is the third phase of cellular respiration.
It takes place in the mitochondria.
In the citric acid cycle, the acetyl group from the preparatory reaction is combined with oxaloacetate to form citrate.
Citrate is then broken down into a series of other molecules, and carbon dioxide is released.
NADH, FADH2, and ATP are also produced during the citric acid cycle.
The Electron Transport Chain
The electron transport chain is the fourth and final phase of cellular respiration.
It takes place in the mitochondria.
In the electron transport chain, electrons from NADH and FADH2 are passed along a series of proteins.
As the electrons move down the chain, energy is released.
This energy is used to pump hydrogen ions across the inner mitochondrial membrane.
The hydrogen ions then flow back down the gradient, and this energy is used to make ATP.
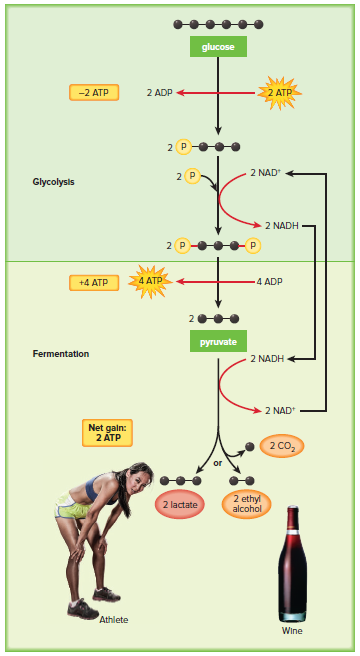
7.2 Outside the Mitochondria: Glycolysis
Glycolysis is a metabolic pathway that takes place in the cytoplasm of both eukaryotes and prokaryotes.
During glycolysis, glucose, a 6-carbon molecule, is broken down to two molecules of pyruvate, a 3-carbon molecule.
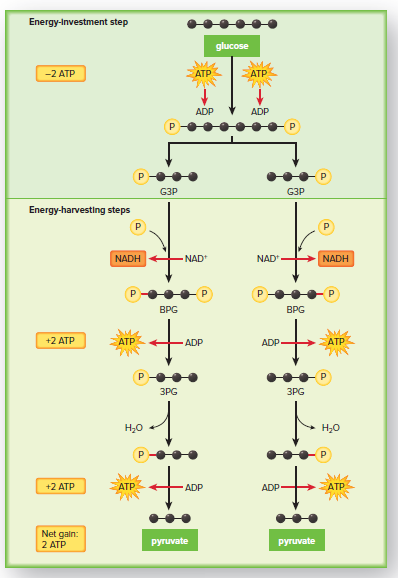
Glycolysis is divided into two phases: the energy-investment phase and the energy-harvesting phase.
In the energy-investment phase, two ATP molecules are used to initiate the reactions of glycolysis.
In the energy-harvesting phase, four ATP molecules are produced, along with two NADH molecules.
The net yield of glycolysis is two ATP molecules and two NADH molecules.
Energy-Investment Step
During the energy-investment step of glycolysis, two ATP molecules transfer their phosphate groups to two different substrates.
This results in the formation of two ADP molecules and two phosphate groups.
The transfer of phosphate groups activates the substrates, making them more reactive.
This allows the substrates to undergo the energy-harvesting reactions of glycolysis.
Energy-Harvesting Steps
During the energy-harvesting steps of glycolysis, electrons and hydrogen ions are removed from the substrates and captured by NAD+, producing two NADH.
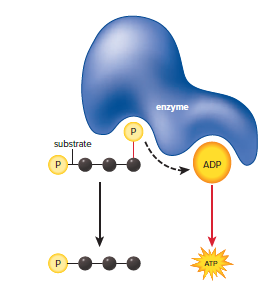
The energized phosphate groups on the intermediates are used to synthesize four ATP through a process called substrate-level ATP synthesis.
The net gain of ATP from glycolysis is 2 ATP.
If oxygen is available, the end product of glycolysis (pyruvate) enters the aerobic reactions within mitochondria, where it is used to generate more ATP.
If oxygen is not available, pyruvate enters the anaerobic fermentation pathways.
In humans, if oxygen is not available, pyruvate is reduced to lactate.
7.3 Outside the Mitochondria Fermentation
The complete breakdown of glucose requires oxygen to keep the electron transport chain working.
If oxygen is limited, pyruvate molecules accumulate in the cell.
Intermediates, such as NAD+ and FAD, cannot be recycled.
Cells may enter anaerobic pathways, such as fermentation, following glycolysis.
There are two basic forms of fermentation: lactic acid and alcohol.
Lactic Acid Fermentation
In animals and some bacteria, pyruvate formed by glycolysis can be reduced to lactate.
This reaction is catalyzed by lactate dehydrogenase, which uses NADH as a source of electrons.
The reduction of pyruvate to lactate regenerates NAD+, which can then be used in the earlier reactions of glycolysis.
This regeneration of NAD+ allows glycolysis to continue, producing ATP by substrate-level phosphorylation.
Anaerobic glycolysis and fermentation are two processes that produce ATP in the absence of oxygen.
These processes only produce a small fraction of the potential energy stored in a glucose molecule.
Despite the low yield, anaerobic pathways are essential for providing a rapid burst of ATP.
Muscle cells are more apt than other cells to carry on fermentation.
When muscles are working vigorously over a short period of time, fermentation is a way to produce ATP even though oxygen is temporarily in limited supply.
When a person stops running, the body is in an oxygen deficit. This means that the body has used up more oxygen than it has taken in.
The body's response to an oxygen deficit is to breathe very heavily. This is called hyperventilation. Hyperventilation helps to increase the amount of oxygen in the blood.
Recovery from an oxygen deficit is complete when the body has taken in enough oxygen to completely break down glucose.
Blood carries away the lactate formed in muscles and transports it to the liver.
In the liver, lactate is converted to pyruvate.
Some of the pyruvate is oxidized completely, and the rest is converted back to glucose.
Alcohol Fermentation
In other organisms (bacteria and fungi), pyruvate is reduced to produce alcohol.
The electrons needed to reduce the pyruvate are supplied by NADH molecules.
NAD+ molecules are regenerated for use in glycolysis.
Alcohol fermentation releases small amounts of CO2.
Yeasts (a type of fungi) are good examples of microorganisms that generate ethyl alcohol and CO2 when they carry out fermentation.
Ethyl alcohol is the desired product when yeasts are used to ferment grapes for wine production or to ferment wort—derived from barley—for beer production.
7.4 Inside the Mitochondria
After glycolysis, if oxygen is present, the cell will enter the aerobic phases of cellular respiration.
The aerobic phases of cellular respiration occur inside the mitochondria.
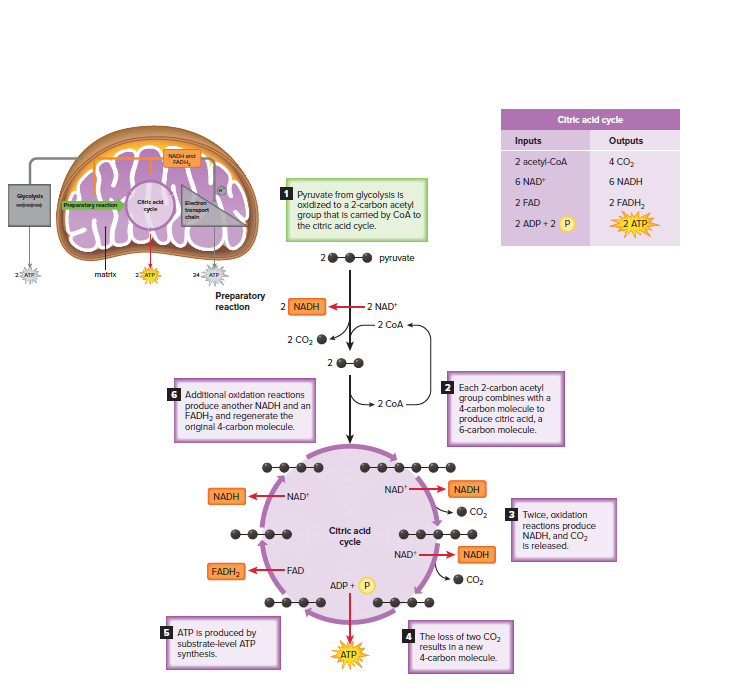
Preparatory Reaction
The preparatory reaction occurs in the matrix of the mitochondria.
It is so named because it prepares the outputs of glycolysis (pyruvate molecules) for use in the citric acid cycle.
Glycolysis splits each glucose into two pyruvate molecules, so the preparatory reaction occurs twice per glucose molecule.
During the preparatory reaction:
Pyruvate is oxidized, and a CO2 molecule is given off.
NAD+ accepts electrons and hydrogen ions, forming NADH.
The product, a 2-carbon acetyl group, is attached to coenzyme A (CoA), forming acetyl-CoA.
Per glucose molecule, the outputs of the preparatory reaction are: 2 CO2, 2 NADH, 2 acetyl-CoA
The Citric Acid Cycle
The citric acid cycle is a cyclical metabolic pathway located in the matrix of mitochondria.
It was originally called the Krebs cycle to honor the scientist who first studied it.
At the start of the citric acid cycle, the 2-carbon acetyl group carried by CoA joins with a 4-carbon molecule, producing a 6-carbon citrate molecule. The CoA is released and is used again in the preparatory reaction.
During the citric acid cycle:
The acetyl group is oxidized, in the process forming CO2.
Both NAD+ and FAD accept electrons and hydrogen ions, resulting in NADH and FADH2.
Substrate-level ATP synthesis occurs, and an ATP results.
Because the citric acid cycle turns twice for each original glucose, the inputs and outputs of the citric acid cycle per glucose are as follows:
The Electron Transport Chain
The electron transport chain is a series of protein complexes that pass electrons from one to another.
NADH and FADH2 deliver electrons to the chain.
The carriers of the electron transport chain accept only electrons and not hydrogen ions.
The electron transport chain is a series of redox reactions that remove the high-energy electrons from NADH and FADH2.
As these reactions occur, energy is released and captured for ATP production.
The final acceptor of electrons is oxygen (O2).
Once NADH has delivered electrons to the electron transport chain, NAD+ is regenerated.
In the same manner, FAD is regenerated.
The recycling of coenzymes increases cellular efficiency.
Generating ATP
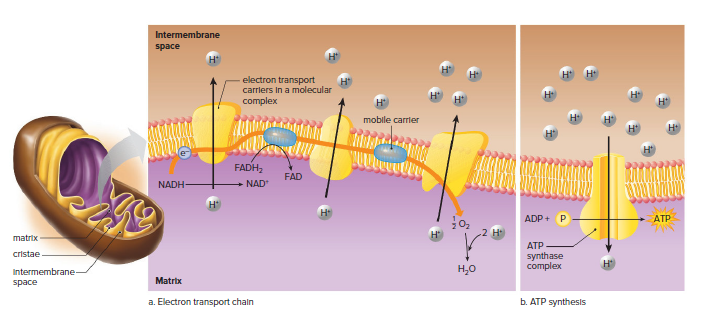
The electron transport chain is a series of protein complexes that transfer electrons from NADH and FADH2 to oxygen.
The electron transport chain is located in the inner mitochondrial membrane.
The energy released by the electron transport chain is used to pump hydrogen ions (H+) from the mitochondrial matrix into the intermembrane space.
The pumping of H+ into the intermembrane space creates a proton gradient.
The proton gradient drives ATP synthase, which uses the energy to produce ATP.
NADH and FADH2 are both produced during cellular respiration, but they do not produce the same amount of ATP.
NADH produces 3 ATP, while FADH2 produces only 2 ATP.
This is because NADH delivers electrons to the first carrier of the electron transport chain, whereas FADH2 delivers its electrons later in the chain.
The H+ gradient is a concentration gradient of hydrogen ions (H+) across the inner mitochondrial membrane.
The H+ gradient contains a large amount of stored energy that can be used to drive forward ATP synthesis.
The ATP synthase complex is an enzyme that uses the energy of the H+ gradient to synthesize ATP from ADP + P.
ATP leaves the matrix by way of a channel protein.
This ATP remains in the cell and is used for cellular work.
7.5 Metabolic Fate of Food
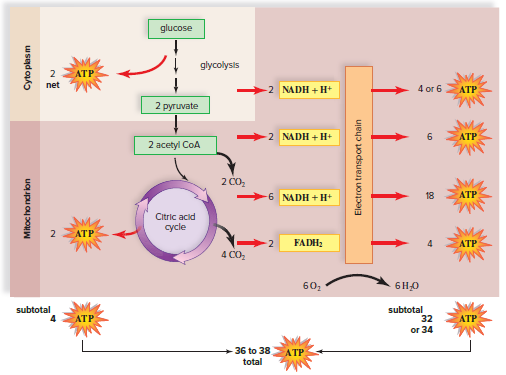
Energy Yield from Glucose Metabolism
Glycolysis produces 2 ATP per glucose molecule.
The citric acid cycle produces 2 ATP per glucose molecule.
Oxidative phosphorylation produces 34 ATP per glucose molecule.
The total ATP yield from the complete breakdown of glucose to CO2 and H2O is 38 ATP.
Electron transport chain and ATP synthase complex: Most of the ATP produced in cells comes from the electron transport chain and the ATP synthase complex.
NADH and FADH2: For each glucose molecule, 10 NADH and 2 FADH2 take electrons from glycolysis and the citric acid cycle to the electron transport chain.
Maximum number of ATP: The maximum number of ATP produced by the electron transport chain is, therefore 34 ATP.
Maximum yield: The maximum number of ATP produced by both the electron transport chain and substrate-level ATP synthesis is 38.
Metabolic differences: Not all cells produce the maximum yield. Metabolic differences cause some cells to produce 36 or fewer ATP.
The yield of 36–38 ATP: A yield of 36–38 ATP represents about 40% of the energy that was initially available in the glucose molecule.
The rest of the energy is lost in the form of heat.
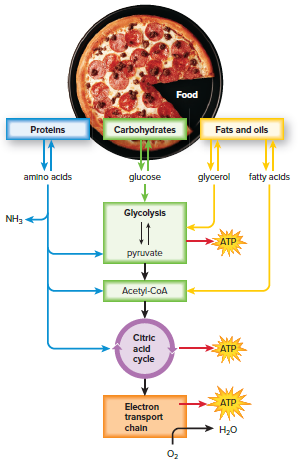
Alternative Metabolic Pathways
Fat breaks down into glycerol and three fatty acid chains when used as an energy source.
Glycerol enters the process of cellular respiration during glycolysis because it is a carbohydrate.
Fatty acids can be metabolized to acetyl groups, which enter the citric acid cycle.
A fatty acid with a chain of 18 carbons can produce three times the number of acetyl groups that a glucose molecule does.
The complete breakdown of glycerol and fatty acids results in more ATP per fat molecule than the breakdown of a glucose molecule.
Only the hydrocarbon backbone of amino acids can be used by the cellular respiration pathways.
The amino group is removed and converted into ammonia (NH3).
Ammonia is then converted into urea, which is the primary excretory product of humans.
The hydrocarbon backbone from an amino acid can enter cellular respiration pathways at pyruvate, acetyl-CoA, or during the citric acid cycle.
Chapter 7: Energy for Cells
7.1 Cellular Respiration
Cellular respiration is the process by which cells produce ATP, the body's main energy source.
ATP is produced by the breakdown of organic molecules, primarily glucose.
Cellular respiration occurs in the mitochondria, the powerhouses of the cell.
Cellular respiration is a redox reaction, which means that electrons are transferred from one molecule to another.
In cellular respiration, glucose is oxidized to form carbon dioxide, and oxygen is reduced to form water.
The energy released from the redox reaction is used to produce ATP.

Phases of Complete Glucose Breakdown
Cellular Respiration
Cellular respiration is a process that cells use to break down glucose and release energy.
The energy released from glucose is used to make ATP, which is the cell's main source of energy.
There are four phases of cellular respiration: glycolysis, the preparatory reaction, the citric acid cycle, and the electron transport chain.
Glycolysis
Glycolysis is the first phase of cellular respiration.
It takes place in the cytoplasm of the cell.
In glycolysis, glucose is broken down into two molecules of pyruvate.
Two ATP molecules are produced during glycolysis.
NADH is also produced during glycolysis.
The Preparatory Reaction
The preparatory reaction is the second phase of cellular respiration.
It takes place in the mitochondria.
In the preparatory reaction, pyruvate is broken down into a 2-carbon acetyl group and carbon dioxide.
NADH is also produced during the preparatory reaction.
The Citric Acid Cycle
The citric acid cycle is the third phase of cellular respiration.
It takes place in the mitochondria.
In the citric acid cycle, the acetyl group from the preparatory reaction is combined with oxaloacetate to form citrate.
Citrate is then broken down into a series of other molecules, and carbon dioxide is released.
NADH, FADH2, and ATP are also produced during the citric acid cycle.
The Electron Transport Chain
The electron transport chain is the fourth and final phase of cellular respiration.
It takes place in the mitochondria.
In the electron transport chain, electrons from NADH and FADH2 are passed along a series of proteins.
As the electrons move down the chain, energy is released.
This energy is used to pump hydrogen ions across the inner mitochondrial membrane.
The hydrogen ions then flow back down the gradient, and this energy is used to make ATP.

7.2 Outside the Mitochondria: Glycolysis
Glycolysis is a metabolic pathway that takes place in the cytoplasm of both eukaryotes and prokaryotes.
During glycolysis, glucose, a 6-carbon molecule, is broken down to two molecules of pyruvate, a 3-carbon molecule.
Glycolysis is divided into two phases: the energy-investment phase and the energy-harvesting phase.
In the energy-investment phase, two ATP molecules are used to initiate the reactions of glycolysis.
In the energy-harvesting phase, four ATP molecules are produced, along with two NADH molecules.
The net yield of glycolysis is two ATP molecules and two NADH molecules.
Energy-Investment Step
During the energy-investment step of glycolysis, two ATP molecules transfer their phosphate groups to two different substrates.
This results in the formation of two ADP molecules and two phosphate groups.
The transfer of phosphate groups activates the substrates, making them more reactive.
This allows the substrates to undergo the energy-harvesting reactions of glycolysis.
Energy-Harvesting Steps
During the energy-harvesting steps of glycolysis, electrons and hydrogen ions are removed from the substrates and captured by NAD+, producing two NADH.
The energized phosphate groups on the intermediates are used to synthesize four ATP through a process called substrate-level ATP synthesis.
The net gain of ATP from glycolysis is 2 ATP.
If oxygen is available, the end product of glycolysis (pyruvate) enters the aerobic reactions within mitochondria, where it is used to generate more ATP.
If oxygen is not available, pyruvate enters the anaerobic fermentation pathways.
In humans, if oxygen is not available, pyruvate is reduced to lactate.


7.3 Outside the Mitochondria Fermentation
The complete breakdown of glucose requires oxygen to keep the electron transport chain working.
If oxygen is limited, pyruvate molecules accumulate in the cell.
Intermediates, such as NAD+ and FAD, cannot be recycled.
Cells may enter anaerobic pathways, such as fermentation, following glycolysis.
There are two basic forms of fermentation: lactic acid and alcohol.
Lactic Acid Fermentation
In animals and some bacteria, pyruvate formed by glycolysis can be reduced to lactate.
This reaction is catalyzed by lactate dehydrogenase, which uses NADH as a source of electrons.
The reduction of pyruvate to lactate regenerates NAD+, which can then be used in the earlier reactions of glycolysis.
This regeneration of NAD+ allows glycolysis to continue, producing ATP by substrate-level phosphorylation.
Anaerobic glycolysis and fermentation are two processes that produce ATP in the absence of oxygen.
These processes only produce a small fraction of the potential energy stored in a glucose molecule.
Despite the low yield, anaerobic pathways are essential for providing a rapid burst of ATP.
Muscle cells are more apt than other cells to carry on fermentation.
When muscles are working vigorously over a short period of time, fermentation is a way to produce ATP even though oxygen is temporarily in limited supply.
When a person stops running, the body is in an oxygen deficit. This means that the body has used up more oxygen than it has taken in.
The body's response to an oxygen deficit is to breathe very heavily. This is called hyperventilation. Hyperventilation helps to increase the amount of oxygen in the blood.
Recovery from an oxygen deficit is complete when the body has taken in enough oxygen to completely break down glucose.
Blood carries away the lactate formed in muscles and transports it to the liver.
In the liver, lactate is converted to pyruvate.
Some of the pyruvate is oxidized completely, and the rest is converted back to glucose.
Alcohol Fermentation
In other organisms (bacteria and fungi), pyruvate is reduced to produce alcohol.
The electrons needed to reduce the pyruvate are supplied by NADH molecules.
NAD+ molecules are regenerated for use in glycolysis.
Alcohol fermentation releases small amounts of CO2.
Yeasts (a type of fungi) are good examples of microorganisms that generate ethyl alcohol and CO2 when they carry out fermentation.
Ethyl alcohol is the desired product when yeasts are used to ferment grapes for wine production or to ferment wort—derived from barley—for beer production.

7.4 Inside the Mitochondria
After glycolysis, if oxygen is present, the cell will enter the aerobic phases of cellular respiration.
The aerobic phases of cellular respiration occur inside the mitochondria.
Preparatory Reaction
The preparatory reaction occurs in the matrix of the mitochondria.
It is so named because it prepares the outputs of glycolysis (pyruvate molecules) for use in the citric acid cycle.
Glycolysis splits each glucose into two pyruvate molecules, so the preparatory reaction occurs twice per glucose molecule.
During the preparatory reaction:
Pyruvate is oxidized, and a CO2 molecule is given off.
NAD+ accepts electrons and hydrogen ions, forming NADH.
The product, a 2-carbon acetyl group, is attached to coenzyme A (CoA), forming acetyl-CoA.
Per glucose molecule, the outputs of the preparatory reaction are: 2 CO2, 2 NADH, 2 acetyl-CoA
The Citric Acid Cycle
The citric acid cycle is a cyclical metabolic pathway located in the matrix of mitochondria.
It was originally called the Krebs cycle to honor the scientist who first studied it.
At the start of the citric acid cycle, the 2-carbon acetyl group carried by CoA joins with a 4-carbon molecule, producing a 6-carbon citrate molecule. The CoA is released and is used again in the preparatory reaction.
During the citric acid cycle:
The acetyl group is oxidized, in the process forming CO2.
Both NAD+ and FAD accept electrons and hydrogen ions, resulting in NADH and FADH2.
Substrate-level ATP synthesis occurs, and an ATP results.
Because the citric acid cycle turns twice for each original glucose, the inputs and outputs of the citric acid cycle per glucose are as follows:

The Electron Transport Chain
The electron transport chain is a series of protein complexes that pass electrons from one to another.
NADH and FADH2 deliver electrons to the chain.
The carriers of the electron transport chain accept only electrons and not hydrogen ions.
The electron transport chain is a series of redox reactions that remove the high-energy electrons from NADH and FADH2.
As these reactions occur, energy is released and captured for ATP production.
The final acceptor of electrons is oxygen (O2).
Once NADH has delivered electrons to the electron transport chain, NAD+ is regenerated.
In the same manner, FAD is regenerated.
The recycling of coenzymes increases cellular efficiency.


Generating ATP
The electron transport chain is a series of protein complexes that transfer electrons from NADH and FADH2 to oxygen.
The electron transport chain is located in the inner mitochondrial membrane.
The energy released by the electron transport chain is used to pump hydrogen ions (H+) from the mitochondrial matrix into the intermembrane space.
The pumping of H+ into the intermembrane space creates a proton gradient.
The proton gradient drives ATP synthase, which uses the energy to produce ATP.
NADH and FADH2 are both produced during cellular respiration, but they do not produce the same amount of ATP.
NADH produces 3 ATP, while FADH2 produces only 2 ATP.
This is because NADH delivers electrons to the first carrier of the electron transport chain, whereas FADH2 delivers its electrons later in the chain.
The H+ gradient is a concentration gradient of hydrogen ions (H+) across the inner mitochondrial membrane.
The H+ gradient contains a large amount of stored energy that can be used to drive forward ATP synthesis.
The ATP synthase complex is an enzyme that uses the energy of the H+ gradient to synthesize ATP from ADP + P.
ATP leaves the matrix by way of a channel protein.
This ATP remains in the cell and is used for cellular work.
7.5 Metabolic Fate of Food
Energy Yield from Glucose Metabolism
Glycolysis produces 2 ATP per glucose molecule.
The citric acid cycle produces 2 ATP per glucose molecule.
Oxidative phosphorylation produces 34 ATP per glucose molecule.
The total ATP yield from the complete breakdown of glucose to CO2 and H2O is 38 ATP.
Electron transport chain and ATP synthase complex: Most of the ATP produced in cells comes from the electron transport chain and the ATP synthase complex.
NADH and FADH2: For each glucose molecule, 10 NADH and 2 FADH2 take electrons from glycolysis and the citric acid cycle to the electron transport chain.
Maximum number of ATP: The maximum number of ATP produced by the electron transport chain is, therefore 34 ATP.
Maximum yield: The maximum number of ATP produced by both the electron transport chain and substrate-level ATP synthesis is 38.
Metabolic differences: Not all cells produce the maximum yield. Metabolic differences cause some cells to produce 36 or fewer ATP.
The yield of 36–38 ATP: A yield of 36–38 ATP represents about 40% of the energy that was initially available in the glucose molecule.
The rest of the energy is lost in the form of heat.
Alternative Metabolic Pathways
Fat breaks down into glycerol and three fatty acid chains when used as an energy source.
Glycerol enters the process of cellular respiration during glycolysis because it is a carbohydrate.
Fatty acids can be metabolized to acetyl groups, which enter the citric acid cycle.
A fatty acid with a chain of 18 carbons can produce three times the number of acetyl groups that a glucose molecule does.
The complete breakdown of glycerol and fatty acids results in more ATP per fat molecule than the breakdown of a glucose molecule.
Only the hydrocarbon backbone of amino acids can be used by the cellular respiration pathways.
The amino group is removed and converted into ammonia (NH3).
Ammonia is then converted into urea, which is the primary excretory product of humans.
The hydrocarbon backbone from an amino acid can enter cellular respiration pathways at pyruvate, acetyl-CoA, or during the citric acid cycle.

 Knowt
Knowt
